 |
| Home | Research | Recent Publications | Personnel | Salomon CV | Publicity | Funding |
 |
| Home | Research | Recent Publications | Personnel | Salomon CV | Publicity | Funding |
| Table of Contents |
|
| Current Projects of the Lipid Research Group |
For descriptions
of current research projects, visit the links below. For general
background information, visit Lipid
Oxidation and Disease. For a brief description of previous work,
visit Key Findings from the Lipid Research
Group.
|
| Lipid Oxidation and Disease |
| In view of
the ready oxidizability of polyunsaturated fatty acids and our aerobic
environment, it is understandable that lipid oxidation plays a key role
in human health. It contributes to normal physiological processes
such as the biosynthesis of the hormone-like prostaglandins, thromboxanes,
and prostacyclins. It may be recruited in the immune response or
programmed cell death (apoptosis), and contributes to changes associated
with aging. It also is involved in pathological processes such as
the ischemia- reperfusion injury associated with heart attacks and stroke,
and may contribute, inter alia, to retinal
degeneration, Alzheimer's, Batten's (juvenile lipofuscinosis), and
Parkinson's diseases (1), as well as amyotrophic lateral sclerosis (2).
Involvement of oxidized phospholipids is suspected in the pathogenesis
of several chronic inflammatory diseases such as antiphospholipid antibody
syndrome (3), rheumatoid arthritis (4, 5), inflammatory bowel disease (6),
and multiple sclerosis (7, 8). However, our understanding of these
processes on a molecular level remains primitive.
The importance and challenge of acheiving a molecular level understanding of these processes is exemplified by the involvement of lipid oxidation in atherogenesis. Three factors contribute to the complexity of the problem: (a) a mixture of fatty esters gives rise to (b) a myriad of electrophilic oxidation products which (c) form covalent adducts with biological nucleophiles. Both the oxidized lipids and their protein adducts may have biological activities that promote atherogenesis. Thus, substantial evidence now suggests that atherosclerotic plaques form when monocytes are recruited into the arterial intima to become macrophages where they become foam cells by accumulating large amounts of oxidized low-density lipoprotein (oxLDL) (9, 10). LDLs are detergent-like globules which transport cholesterol and lipids in blood. Their outer hydrophilic shell is composed mainly of phospholipids, free cholesterol, and a protein (apoB-100), while their lipophilic core contains cholesterol esters, and triglycerides. Prominent during the oxidation of LDL is the disappearance of linoleic acid (LA) and arachidonic acid (AA) esters and the formation of numerous reactive oxidation products, many of which attach themselves to the LDL protein. While the biological importance of oxLDL is known, the molecular details of the LDL to oxLDL transformation are not fully understood. In fact, "oxLDL is not a single defined chemical entity. Depending on the extent of oxidation and on the various oxidized products generated, it may show a broad spectrum of biological effects" (11). |
|
|
|
|
|
| Key Findings from the Lipid Research Group |
| Index: Levuglandins / Isolevuglandins / Carboxyalkylpyrroles / Hydroxyoxoalkenoyl Phospholipids |
|
|
| (1) | Salomon, R. G. and Salomon, M. F. (1977) 2,3-Dioxabicyclo[2.2.1]heptane. The strained bicyclic peroxide nucleus of prostaglandin endoperoxides. J. Am. Chem. Soc. 99 3501-3. |
| (2) | Salomon, R. G., Miller, D. B., Zagorski, M. G. and Coughlin, D. J. (1984) Prostaglandin endoperoxides. 14. Solvent-induced fragmentation of prostaglandin endoperoxides. New aldehyde products from PGH2 and a novel intramolecular 1,2-hydride shift during endoperoxide fragmentation in aqueous solution. J. Am. Chem. Soc. 106 6049-60. |
| (3) | Salomon, R. G., Jirousek, M. R., Ghosh, S. and Sharma, R. B. (1987) Prostaglandin endoperoxides. 21. Covalent binding of levuglandin E2 with proteins. Prostaglandins 34 643-56. |
| (4) | Iyer, R. S., Ghosh, S. and Salomon, R. G. (1989) Levuglandin E2 crosslinks proteins. Prostaglandins 37 471-80. |
| (5) | Murthi, K. K., Friedman, L. R., Oleinick, N. L. and Salomon, R. G. (1993) Formation of DNA-protein cross-links in mammalian cells by levuglandin E2. Biochemistry 32 4090-7. |
| (6) | Salomon, R. G., Subbanagounder, G., O'Neil, J., Kaur, K., Smith, M. A., Hoff, H. F., Perry, G. and Monnier, V. M. (1997) Levuglandin E2-Protein Adducts in Human Plasma and Vasculature. Chem. Res. Toxicol. 10 536-545. |
| (7) | Boutaud, O., Brame, C. J., Salomon, R. G., Roberts, L. J., II and Oates, J. A. (1999) Characterization of the Lysyl Adducts Formed from Prostaglandin H2 via the Levuglandin Pathway. Biochemistry 38 9389-9396. |
| (8) | Brame, C. J., Salomon, R. G., Morrow, J. D. and Roberts, L. J., II (1999) Identification of extremely reactive g-ketoaldehydes (isolevuglandins) as products of the isoprostane pathway and characterization of their lysyl protein adducts. J. Biol. Chem. 274 13139-13146. |
|
|
|
|
| (1) | Salomon, R. G., Subbanagounder, G., Singh, U., O'Neil, J. and Hoff, H. F. (1997) Oxidation of Low-Density Lipoproteins Produces Levuglandin-Protein Adducts. Chem. Res. Toxicol. 10 750-759. |
| (2) | Nugteren, D. H., Vonkeman, H. and Dorp, D. A. v. (1967) Non-enzymic conversion of all-cis 8,11,14-eicosatrienoic acid into prostaglandin E1. Rec. Trav. Chim. 86 1237-1245. |
| (3) | Morrow, J. D., Harris, T. M. and L. J. Roberts, I. (1990) Noncyclooxygenase oxidative formation of a series of novel prostaglandins: analytical ramifications for measurement of eicosanoids. Anal. Biochem. 184 1-10. |
| (4) | Morrow, J. D., Hill, K. E., Burk, R. F., Nammour, T. M., Badr, K. F. and L. J. Roberts, I. (1990) A series of prostaglandin F2-like compounds are produced in vivo in humans by a non-cyclooxygenase, free radical-catalyzed mechanism. Proc. Natl. Acad. Sci. USA 87 9383-9387. |
| (5) | Salomon, R. G., Batyreva, E., Kaur, K., Sprecher, D. L., Schreiber, M. J., Crabb, J. W., Penn, M. S., DiCorletoe, A. M., Hazen, S. L. and Podrez, E. A. (2000) Isolevuglandin-protein adducts in humans: products of free radical- induced lipid oxidation through the isoprostane pathway. Biochim Biophys Acta 1485 225-35. |
| (6) | Salomon, R. G., Kaur, K. and Batyreva, E. (2000) Isolevuglandin-protein adducts in oxidized low density lipoprotein and human plasma. A strong connection with cardiovascular disease. Trends Cardiovasc Med 10 53-9. |
|
|
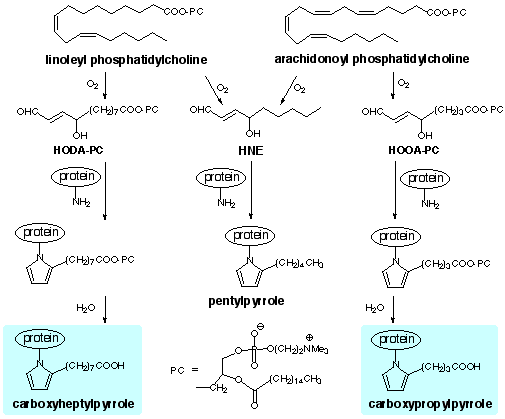 |
| (1) | Esterbauer, H., Schaur, R. J. and Zollner, H. (1991) Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radical Biol. Med. 11 81-128. |
| (2) | Sayre, L. M., Arora, P. K., Iyer, R. S. and Salomon, R. G. (1993) Pyrrole Formation from 4-Hydroxynonenal and Primary Amines. Chem. Res. Toxicol. 6 19-22. |
| (3) | Kaur, K., Salomon, R. G., O'Neil, J. and Hoff, H. F. (1997) Carboxyalkyl Pyrroles in Human Plasma and Oxidized Low Density Lipoproteins. Chem. Res. Toxicol. 10 1387-1396. |
| (4) | J. G. Hollyfield, V. L. Bonilha, M. E. Rayborn, X. Yang, K. G. Shadrach, L. Lu, R. L. Ufret, R. G. Salomon and V. L. Perez "Oxidative damage-induced inflammation initiates age-related macular degeneration", Nat Med 2008, 14, 194-8. |
| (5) | S. L. Doyle, M. Campbell, E. Ozaki, R. G. Salomon, A. Mori, P. Kenna, G. J. Farrar, A. S. Kiang, M. Humphries, E. C. Lavelle, L. A. O'Neill, J. G. Hollyfield and P. Humphries "NLRP3 has a protective role in age-related macular degeneration through the induction of IL-18 by drusen components", Nat Med 2012, 18, 791-8. |
| (6) | F. Cruz-Guilloty, A. M. Saeed, J. J. Echegaray, S. Duffort, A. Ballmick, Y. Tan, M. Betancourt, E. Viteri, G. C. Ramkhellawan, E. Ewald, W. Feuer, D. Huang, R. Wen, L. Hong, H. Wang, J. M. Laird, A. Sene, R. S. Apte, R. G. Salomon, J. G. Hollyfield and V. L. Perez "Infiltration of proinflammatory m1 macrophages into the outer retina precedes damage in a mouse model of age-related macular degeneration", Int J Inflam 2013, 2013, 503725, PMCID 3606733. |
|
|
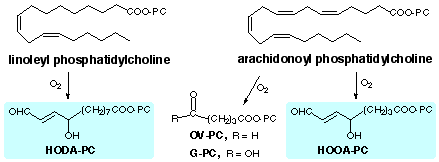 |
| (1) | Watson, A. D.; Leitinger, N.; Navab, M.; Faull, K. F.; Hörkkö; Witztum, J. L.; Palinski, W.; Schwenke, D.; Salomon, R. G.; Sha, W.; Subbanagounder, G.; Fogelman, A. M.; Berliner, J. A. (1997) Structural Identification by Mass Spectrometry of Oxidized Phospholipids in Minimally Oxidized Low Density Lipoprotein That Induce Monocyte/Endothelial Interactions and Evidence for Their Presence in Vivo., J. Biol. Chem., 272, 13597-13607 |
|
|
|
|
| Home | Research | Publications | Personnel | Salomon CV | Publicity | Funding |