First-ever freeze-dried artificial platelets are shelf-stable and portable—a major advance for field medicine
Patients in danger of bleeding out from trauma could be treated on site
Platelets, which allow blood to clot, can save patients in danger of bleeding to death from traumatic injuries. But donated platelets can only be given to patients in a hospital and last only a few days at room temperature to a few weeks when refrigerated.
In a major advance for trauma medicine, researchers at Case Western Reserve University, the University of Pittsburgh and Haima Therapeutics have demonstrated that synthetic platelets can be freeze-dried and remain stable and effective for at least a year at room temperature and at least two months at high temperatures.

“You don’t need a refrigerator to store it, and in a powder form it can be transported to and stockpiled wherever it’s needed,” said Anirban Sen Gupta, the Wallace R. Persons Professor of Biomedical Engineering at Case Western Reserve University School of Medicine and Case School of Engineering, who led the study. “Platelets currently cannot be taken outside of a hospital. But if you could give platelets closer to the point of injury, you improve the chance of survival.”
This unlocks the potential for administering synthetic platelets on a battlefield or on site at a car accident or mass casualty event, which may save more lives, said Sen Gupta, also chief technology officer of Haima, a company he cofounded with CWRU alumna Christa Pawlowski that is developing and commercializing the synthetic platelets.
The team’s findings are published in the journal Advanced Science.

“Developing a freeze-dried formulation is a critical milestone in translating this technology from the lab to real-world use,” said Pawlowski, Haima’s chief operating officer and executive vice president of research and development. “A shelf-stable, portable product is essential for scalable manufacturing, distribution and stockpiling, and it significantly expands the ability to use the product to save lives both in the hospital and in prehospital settings.”
Successful direct injection of synthetic platelets into bone marrow
The researchers also successfully tested a novel technique in laboratory studies, injecting synthetic platelets directly into the bone marrow. This technique could provide a faster way to treat people experiencing life-threatening bleeding in remote areas or disaster zones, where establishing a traditional intravenous (IV) line may pose logistical challenges. Research for this approach was published recently in the Journal of Thrombosis and Haemostasis.
The synthetic platelets were manufactured, freeze-dried and tested by Haima, with further testing by researchers at Case Western Reserve and Pitt's School of Medicine. Matthew Neal, Watson Chair and Professor of Surgery, led the team at Pitt and is chief medical officer at Haima.

“On the battlefield or at the scene of a car accident, it can be impossible to secure an IV line to deliver blood through the vein of a severely bleeding victim,” said Neal, who is also co-director of the Trauma and Transfusion Medicine Research Center at Pitt. “That is why it was critical that we ensure this life-saving synthetic platelet technology be effective when delivered directly into bone, one of the most frequent delivery routes used in prehospital resuscitation.”
Freeze-dried platelets remain effective after extreme temps and long storage
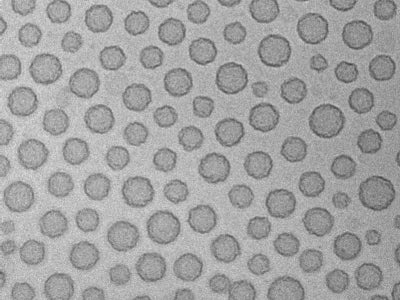
To study the stability and effectiveness of the freeze-dried synthetic platelets, the researchers subjected the nanoparticles to extreme temperatures and lengthy storage. They rehydrated the particles and tested their effectiveness.
They found that the synthetic platelets remained effective after a year of being stored at room temperature, and two months at 50-degrees Celsius—over 100 degrees Fahrenheit—which means they could be taken to locations where cold storage isn’t readily available, without losing their potency.
Direct injection could save lives in the field
After consulting with trauma medics, Sen Gupta and his team learned that starting an IV can be extremely difficult during severe bleeding emergencies, particularly in challenging environments like battlefields or remote locations. This led them to test a more direct approach: injecting the synthetic platelet nanoparticles directly into the shin, where the bone sits close to the skin’s surface.
It is similar in concept to an EpiPen that’s jabbed into a person’s thigh to prevent a severe allergic reaction. A medical professional can quickly jab a needle into the marrow of the bone and deliver the treatment without having to establish an IV line. The research demonstrated that this technique allows the nanoparticles to rapidly enter the bloodstream, travel to the site of injury and help stop bleeding.
This has the added advantage of speed in a situation where shaving seconds could make the difference between life and death.
Support and collaboration
The research was supported by U.S. Department of Defense (DoD) grants to Sen Gupta, Neal and Haima.
Sen Gupta and Pawlowski developed the synthetic platelets at Case Western Reserve and licensed the technology to Haima. Haima has recently received two Small Business Innovation Research grants from the Defense Advanced Research Projects Agency (DARPA) to manufacture and test the synthetic platelet product, SynthoPlate, in animal models for safety. The company hopes to apply to the U.S. Food and Drug Administration to begin human clinical trials in 2027.