Ensuring Regulatory Compliance and Research Integrity
At Case Western Reserve, the Office of Research Compliance (ORC) helps researchers stay compliant with federal, state and local regulations throughout the research lifecycle. Explore the Office of Research and Technology Management’s educational opportunities or the components of regulatory compliance oversight below.

Research Oversight
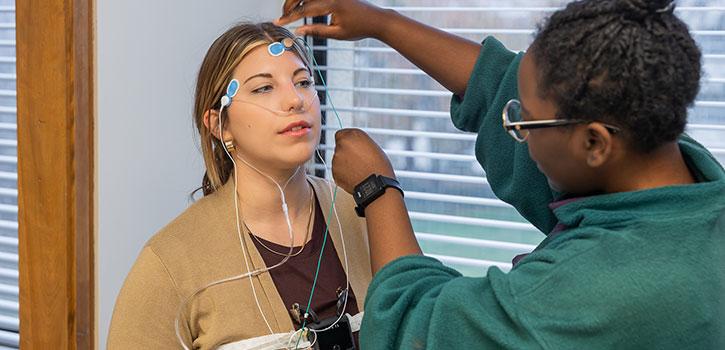
Human Subjects Research
The Human Research Protection Program (HRPP) reviews and monitors human research to ensure the appropriate steps are taken to protect the rights and welfare of research participants.

Animal Subjects Research
The Institutional Animal Care and Use Committee (IACUC) provides assistance to investigators in fulfilling their obligation to plan and conduct animal experiments in accord with the highest scientific, humane and ethical principles.

Conflict of Interests
The Conflict of Interests (COI) Committee aims to preserve the integrity of the university and maintain compliance with applicable federal regulations, while respecting academic freedom and encouraging outside scholarly and entrepreneurial activities.
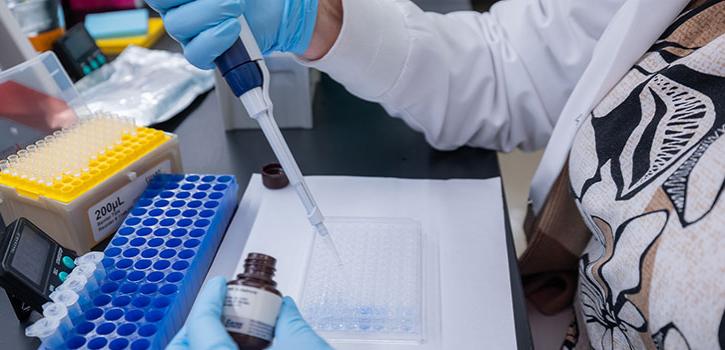
Biosafety
The Institutional Biosafety Committee (IBC) supports research with recombinant DNA (rDNA) and synthetic nucleic acid molecules.

Human Stem Cells
The Human Stem Cell Research Oversight (HSCRO) Committee provides oversight and guidance for research involving human pluripotent stem cells.

Dual Use Research of Concern (DURC)
The university's Institutional Review Entity (IRE) Committee provides oversight for Dual Use Research of Concern (DURC), or research involving agents that have the potential to threaten public health and safety.
Research Integrity
CWRU is committed to the integrity of all research conducted under its purview. All observed, suspected or apparent research misconduct should be reported to the research integrity officer.
If anyone associated with CWRU has a concern about the integrity of research, there are resources to provide information to the appropriate officials so that the situation can be reviewed.



